“If we knew what we were doing, it would not be called research”
Albert Einstein
Our Research |
Donor-Unrestricted T cells (DURTs)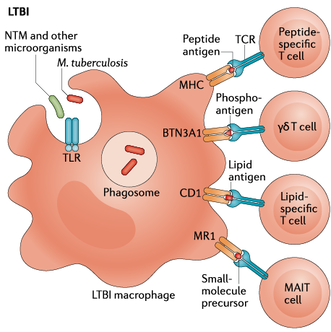 Figure reproduced from Simmons et al. Nature Reviews Immunology 2018 (PMID 29895826) Figure reproduced from Simmons et al. Nature Reviews Immunology 2018 (PMID 29895826)
Peptide antigens are typically presented to T cells by highly polymorphic major histocompatibility complex (MHC) Class I and Class II molecules and have been studied extensively in new vaccines against tuberculosis. However, T cells can also be activated by non-peptide antigens via non-polymorphic and MHC-independent antigen presenting systems. These T cells exhibit conserved features of antigen recognition across genetically unrelated donors, leading to their designation as ‘donor-unrestricted’ T cells (DURTs). Our lab is studying the role of DURTs in mediating protective immunity against M. tuberculosis.
|
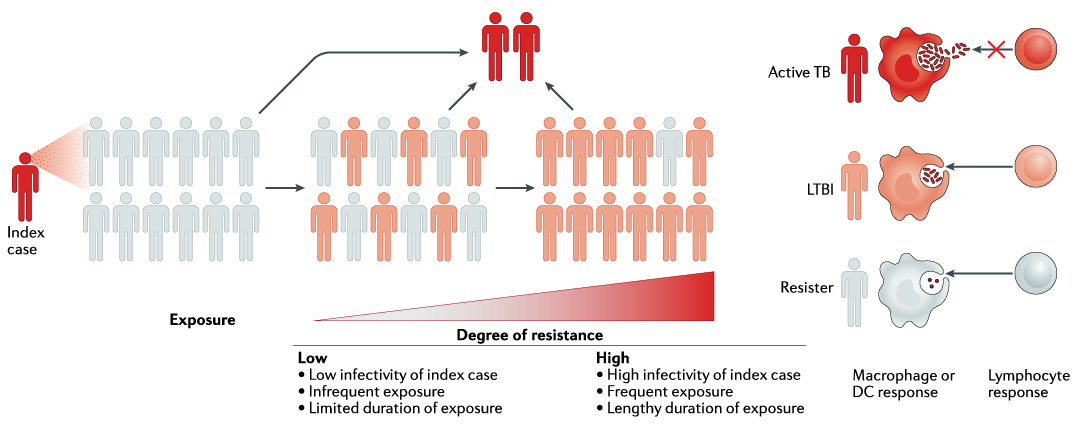
Resistance to M. tuberculosis |
|
In TB endemic settings, some individuals who are heavily and repeatedly exposed to M. tuberculosis never show evidence of immune sensitization as measured by a tuberculin skin test (TST) or IFN-γ release assay (IGRA). We hypothesize that these individuals may “resist” M.tb infection. We are part of a large consortium investigating the immune mechanisms underlying this poorly understood but potentially important clinical observation. Our findings may inform the development of new therapeutic approaches (vaccines and host-directed therapies) for TB.
|
Modeling Human TB GranulomasGranulomas are formed in response to persistent immune stimulation by pathogens through aggregation of immune cells into organized structures. Studies have demonstrated both protective and pathologic properties of granulomas in TB. However, research has been limited by the ability of current cell culture and animal models to fully capture the features of human granulomas. We are working to develop better in-vitro models of human TB granulomas using microfluidics and lymphoid organoids.
Controlled Human Infection with Mycobacteria (CHIM) Human challenge models have greatly accelerated vaccine development for malaria, typhoid, and dengue among other infectious diseases. The development of new vaccines and drugs for TB would benefit from a human challenge model because efficacy trials are long and expensive for relevant clinical endpoints (e.g. symptomatic pulmonary TB). However, significant challenges have impeded progress including the myriad clinical outcomes that result from primary infection with M. tuberculosis, limitations in the sensitivity of current diagnostics, and the ethics of challenging humans with virulent mycobacteria. In collaboration with investigators at UW and Fred Hutch Cancer Center, we are working to establish a tractable CHIM model using BCG to probe in-vivo host-pathogen biology and evaluate new treatments for TB.
|